NEWS ABOUT THE PERIODIC TABLE:
(August 31, 2013)
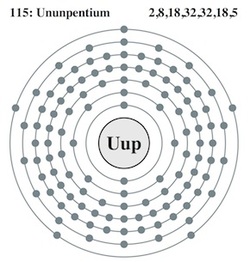
Ununpentium, the Newest Element
Posted by Michael Lemonick
When the Russian chemist Dmitri Mendeleev published his periodic table of elements in 1869, there were just fifty-nine entries on it. The table grouped those elements—hydrogen, oxygen, and carbon, along with less familiar substances like osmium, rhodium, yttrium—according to their shared chemical properties and the weight of their atoms. The table also contained thirty-three empty spaces that implied that there were elements still to be discovered.
Illustration by Greg Robson/Pumbaa
What Mendeleev couldn’t have imagined was that scientists would one day begin creating elements not found naturally. Just a few days ago, the fabrication of one of the heaviest elements yet was confirmed by Swedish scientists working at the G.S.I. Helmholtz Center for Heavy Ion Research, in Darmstadt, Germany. Its provisional name is ununpentium.
What makes an element distinct is the number of protons it has in its nucleus: hydrogen has one proton, helium has two, and on up the periodic table to uranium, which has ninety-two. Creating new elements began with physicists bombarding existing ones with other particles; as the nuclei careened around they would sometimes smash together and form atoms with more than ninety-two protons. First came neptunium, in 1940, with ninety-three protons, then plutonium, with ninety-four (which, it turns out, does exist in trace quantities in nature). In the years since, scientists continued creating heavier and generally more unstable atoms. Ununpentium element has a hundred and fifteen protons. (It’s name means “one-one-five.”)
A few artificial elements have important practical applications. Plutonium can be used in nuclear weapons; it’s also the fuel for some nuclear reactors and has been used to power space probes, including Voyager 1 and 2. Ununpentium has no practical uses yet. It’s so unstable that it doesn’t stay around long enough to make anything out of it. Almost immediately after the Swedish scientists created it by smashing twenty-proton calcium nuclei into ninety-five-proton americium nuclei at high speed, the ununpentium decayed into element 113—ununtrium—which itself decayed into lighter elements. Its half-life was found to be only a hundred and seventy-three milliseconds. But the decay chain, plus the X-rays and gamma rays the short-lived nuclei spat out in their death throes, convinced the physicists that the one that got away was indeed element 115.
In fact, this was the second sighting of the element: Russian scientists had claimed the discovery of element 115 back in 2003, but the International Union of Pure and Applied Chemistry—chemistry’s equivalent of the International Astronomical Union, which famously demoted Pluto from planet status in 2006—wouldn’t acknowledge it without a confirming experiment from another team.
Ununpentium’s brief life doesn’t mean it’s completely useless: nuclear physicists can try and deduce from properties more about how atomic nuclei are put together and how they fall apart. The latest experiment created about thirty atoms’ worth of element 115; physicists will continue trying to make larger batches in order to explore its properties more fully.
Ununpentium is not, however, the heaviest known atom. In general, it’s harder to make new elements the further up the periodic table you go. But owing to the complex structure of heavy nuclei, atoms more massive than ununpentium were created earlier. I.U.P.A.C. has already signed off on element 116 (livermorium), element 117 (ununseptium) and element 118 (ununoctium), although the latter two haven’t been assigned permanent names yet. Ununoctium’s half life is just 0.89 milliseconds.
While it’s generally true that heavier atoms are more unstable than lighter ones, there’s at least the possibility, first proposed by the physicist Glenn Seaborg back in the nineteen-sixties, that if an atom gets heavy enough it could arrive at an “island of stability,” far less prone to immediate decay than its neighbors on the periodic table. Unbinilium, for example—the still-theoretical element 120—might be one of them. Perhaps it could live for minutes, or even days. That’s still far too short a time for almost any practical use. But for scientists who study nuclei for a living, the prospect of keeping an element that heavy around for more than a handful of milliseconds is an almost unimaginable luxury.
Source : http://www.newyorker.com/online/blogs/elements/2013/08/unumpentium-the-new-artificial-element.html
SUBMITTED BY: KIMBERLY VELASCO (8-AVOCADO)
-SHARYN RAMIREZ
Ununpentium, the Newest Element
Posted by Michael Lemonick
When the Russian chemist Dmitri Mendeleev published his periodic table of elements in 1869, there were just fifty-nine entries on it. The table grouped those elements—hydrogen, oxygen, and carbon, along with less familiar substances like osmium, rhodium, yttrium—according to their shared chemical properties and the weight of their atoms. The table also contained thirty-three empty spaces that implied that there were elements still to be discovered.
Illustration by Greg Robson/Pumbaa
What Mendeleev couldn’t have imagined was that scientists would one day begin creating elements not found naturally. Just a few days ago, the fabrication of one of the heaviest elements yet was confirmed by Swedish scientists working at the G.S.I. Helmholtz Center for Heavy Ion Research, in Darmstadt, Germany. Its provisional name is ununpentium.
What makes an element distinct is the number of protons it has in its nucleus: hydrogen has one proton, helium has two, and on up the periodic table to uranium, which has ninety-two. Creating new elements began with physicists bombarding existing ones with other particles; as the nuclei careened around they would sometimes smash together and form atoms with more than ninety-two protons. First came neptunium, in 1940, with ninety-three protons, then plutonium, with ninety-four (which, it turns out, does exist in trace quantities in nature). In the years since, scientists continued creating heavier and generally more unstable atoms. Ununpentium element has a hundred and fifteen protons. (It’s name means “one-one-five.”)
A few artificial elements have important practical applications. Plutonium can be used in nuclear weapons; it’s also the fuel for some nuclear reactors and has been used to power space probes, including Voyager 1 and 2. Ununpentium has no practical uses yet. It’s so unstable that it doesn’t stay around long enough to make anything out of it. Almost immediately after the Swedish scientists created it by smashing twenty-proton calcium nuclei into ninety-five-proton americium nuclei at high speed, the ununpentium decayed into element 113—ununtrium—which itself decayed into lighter elements. Its half-life was found to be only a hundred and seventy-three milliseconds. But the decay chain, plus the X-rays and gamma rays the short-lived nuclei spat out in their death throes, convinced the physicists that the one that got away was indeed element 115.
In fact, this was the second sighting of the element: Russian scientists had claimed the discovery of element 115 back in 2003, but the International Union of Pure and Applied Chemistry—chemistry’s equivalent of the International Astronomical Union, which famously demoted Pluto from planet status in 2006—wouldn’t acknowledge it without a confirming experiment from another team.
Ununpentium’s brief life doesn’t mean it’s completely useless: nuclear physicists can try and deduce from properties more about how atomic nuclei are put together and how they fall apart. The latest experiment created about thirty atoms’ worth of element 115; physicists will continue trying to make larger batches in order to explore its properties more fully.
Ununpentium is not, however, the heaviest known atom. In general, it’s harder to make new elements the further up the periodic table you go. But owing to the complex structure of heavy nuclei, atoms more massive than ununpentium were created earlier. I.U.P.A.C. has already signed off on element 116 (livermorium), element 117 (ununseptium) and element 118 (ununoctium), although the latter two haven’t been assigned permanent names yet. Ununoctium’s half life is just 0.89 milliseconds.
While it’s generally true that heavier atoms are more unstable than lighter ones, there’s at least the possibility, first proposed by the physicist Glenn Seaborg back in the nineteen-sixties, that if an atom gets heavy enough it could arrive at an “island of stability,” far less prone to immediate decay than its neighbors on the periodic table. Unbinilium, for example—the still-theoretical element 120—might be one of them. Perhaps it could live for minutes, or even days. That’s still far too short a time for almost any practical use. But for scientists who study nuclei for a living, the prospect of keeping an element that heavy around for more than a handful of milliseconds is an almost unimaginable luxury.
Source : http://www.newyorker.com/online/blogs/elements/2013/08/unumpentium-the-new-artificial-element.html
SUBMITTED BY: KIMBERLY VELASCO (8-AVOCADO)
-SHARYN RAMIREZ
Meet 115, the Newest Element on the Periodic Table
The extremely heavy element was just confirmed by scientists in Sweden.
Periodic Table of Elements
PERIODIC TABLE COURTESY OF TOMACCO/GETTY IMAGES
BY Melody Kramer
for National Geographic
PUBLISHED AUGUST 28, 2013
If you've learned all the elements from actinium to zirconium, it's time to head back to the periodic table, where there's a new, extremely heavy element in town.
The new element doesn't have an official name yet, so scientists are calling it ununpentium, based on the Latin and Greek words for its atomic number, 115. (Related: Read a feature on element hunters in National Geographic magazine.)
In case you forgot your high school chemistry, here's a quick refresher: An element's atomic number is the number of protons it contains in its nucleus.
The heaviest element in nature is uranium, which has 92 protons. But heavier elements-which have more protons in their nucleus-can be created through nuclear fusion. (Related: Learn how to make an element.)
The man-made 115 was first created by Russian scientists in Dubna about ten years ago. This week, chemists at Lund University in Sweden announced that they had replicated the Russian study at the GSI Helmholtz Center for Heavy Ion Research in Germany.
Element 115 will join its neighbors 114 and 116-flerovium and livermorium, respectively-on the periodic table just as soon as a committee from the International Union of Pure and Applied Chemistry(IUPAC) decides on an official name for 115.
We asked Paul Hooker, a chemistry professor at Westminster College in Salt Lake City, Utah, for his take on the latest addition to the periodic table.
So it sounds like 115 was actually created ten years ago, by a lab in Russia. Why are we just learning about its discovery?
When you find a new element, it has to be confirmed. You need two different labs to confirm it before [IUPAC] considers adding it to the periodic table.
This is the second lab coming in and repeating the same experiment, so now it's considered to be an official new element.
So what did the Russian and Swedish chemists actually do?
The way that you make new elements now is by shooting a beam of an element at another element and then seeing what happens when they collide.
In this case, the researchers used americium, which is kind of interesting because it's an unstable, radioactive element. They fired calcium atoms-which are much lighter than americium atoms-at the americium for weeks or even months. Most of the calcium atoms bounced off, but every now and then the atoms collided and instead of the calcium element bouncing off, it actually stuck to the americium element. When that happens, you get a short-lived atom with more protons in its nucleus, which is the center of the new element 115.
How did they know they created a new element if it happened so quickly? I think I read that it existed for less than a second before it decayed.
They look for the decay products. They look for telltale signs for when 115 disintegrates, by what's called alpha particle emission. When they see enough of those signals, they can say they probably formed a new element.
How do they know if a new element will be unstable or not?
There was an element 118 that was predicted to be much more stable; 115 wasn't predicted to be especially stable. We know what is stable. Certain ratios of protons to neutrons are stable. As the nucleus gets bigger and bigger, it's not stable-and then it can radioactively decay and spit out smaller particles-that means it's really not very stable.
Can anyone try to create a new element?
No. You need a large vacuum chamber because you can't fire calcium atoms through the air. You need a lot of specialized equipment. There aren't many labs that can do this type of thing. The only people interested in doing this are trying to answer some of the bigger questions, like "How is all matter held together?"
Where are most new elements created?
Most of these new elements have been formed in Russia and the States for the past 30 to 40 years. It's become a race for who can get the next new element, to try to make the biggest one you actually can. But of course, because they're so big, they're very unstable and fall apart extremely quickly.
If they fall apart extremely quickly-and clearly don't exist in nature-then what's the point?
I talk about this a lot with my students. I basically tell them, "Because it's there." There's no way that a new, unstable element is going to have any uses because it deteriorates so quickly. But it gives insight about the forces that hold atoms together so we can learn more about how the universe is held together.
Why are people really doing this? Why do we send particles through huge colliders? Why are we smashing things into each other at higher and higher velocities? I think it fulfills the human race's natural curiosity. We want to know where we come from. And every time we answer something, we come up with ten more questions to answer.
This was a man-made element. How do we know we've found all of the naturally occurring elements?
The good thing with elements is that they're defined by atomic numbers, meaning they're defined by the number of protons in the nucleus. This number is never a fraction, so you can't have, say, 3.2 protons in a nucleus. So we know we have them all because we know of an element with one proton and an element with two protons and so on.
Is there a limit to how many elements we can create?
Well, we're hitting a limit with stability when there are over 90 protons in a nucleus, so while we may find more, we're certainly not getting up to 1,000 protons. It would be too unstable.
One last question: I actually have a periodic table shower curtain. Do you recommend getting an updated one?
I recommend updating your shower curtain when 115 is confirmed. When the committee gets together and names it. And that's an entirely different question.
So I guess I should ask: Why does a committee do it?
Because these things get quite political. Back in the day, the Americans would say: We discovered it and named it something. The Russians would say: We did, and named it something else. So a committee has to get together and negotiate. They try to keep it apolitical-maybe they'll name it after someone from Italy or Lithuania or something.
-GLAIZA GANGAN
The extremely heavy element was just confirmed by scientists in Sweden.
Periodic Table of Elements
PERIODIC TABLE COURTESY OF TOMACCO/GETTY IMAGES
BY Melody Kramer
for National Geographic
PUBLISHED AUGUST 28, 2013
If you've learned all the elements from actinium to zirconium, it's time to head back to the periodic table, where there's a new, extremely heavy element in town.
The new element doesn't have an official name yet, so scientists are calling it ununpentium, based on the Latin and Greek words for its atomic number, 115. (Related: Read a feature on element hunters in National Geographic magazine.)
In case you forgot your high school chemistry, here's a quick refresher: An element's atomic number is the number of protons it contains in its nucleus.
The heaviest element in nature is uranium, which has 92 protons. But heavier elements-which have more protons in their nucleus-can be created through nuclear fusion. (Related: Learn how to make an element.)
The man-made 115 was first created by Russian scientists in Dubna about ten years ago. This week, chemists at Lund University in Sweden announced that they had replicated the Russian study at the GSI Helmholtz Center for Heavy Ion Research in Germany.
Element 115 will join its neighbors 114 and 116-flerovium and livermorium, respectively-on the periodic table just as soon as a committee from the International Union of Pure and Applied Chemistry(IUPAC) decides on an official name for 115.
We asked Paul Hooker, a chemistry professor at Westminster College in Salt Lake City, Utah, for his take on the latest addition to the periodic table.
So it sounds like 115 was actually created ten years ago, by a lab in Russia. Why are we just learning about its discovery?
When you find a new element, it has to be confirmed. You need two different labs to confirm it before [IUPAC] considers adding it to the periodic table.
This is the second lab coming in and repeating the same experiment, so now it's considered to be an official new element.
So what did the Russian and Swedish chemists actually do?
The way that you make new elements now is by shooting a beam of an element at another element and then seeing what happens when they collide.
In this case, the researchers used americium, which is kind of interesting because it's an unstable, radioactive element. They fired calcium atoms-which are much lighter than americium atoms-at the americium for weeks or even months. Most of the calcium atoms bounced off, but every now and then the atoms collided and instead of the calcium element bouncing off, it actually stuck to the americium element. When that happens, you get a short-lived atom with more protons in its nucleus, which is the center of the new element 115.
How did they know they created a new element if it happened so quickly? I think I read that it existed for less than a second before it decayed.
They look for the decay products. They look for telltale signs for when 115 disintegrates, by what's called alpha particle emission. When they see enough of those signals, they can say they probably formed a new element.
How do they know if a new element will be unstable or not?
There was an element 118 that was predicted to be much more stable; 115 wasn't predicted to be especially stable. We know what is stable. Certain ratios of protons to neutrons are stable. As the nucleus gets bigger and bigger, it's not stable-and then it can radioactively decay and spit out smaller particles-that means it's really not very stable.
Can anyone try to create a new element?
No. You need a large vacuum chamber because you can't fire calcium atoms through the air. You need a lot of specialized equipment. There aren't many labs that can do this type of thing. The only people interested in doing this are trying to answer some of the bigger questions, like "How is all matter held together?"
Where are most new elements created?
Most of these new elements have been formed in Russia and the States for the past 30 to 40 years. It's become a race for who can get the next new element, to try to make the biggest one you actually can. But of course, because they're so big, they're very unstable and fall apart extremely quickly.
If they fall apart extremely quickly-and clearly don't exist in nature-then what's the point?
I talk about this a lot with my students. I basically tell them, "Because it's there." There's no way that a new, unstable element is going to have any uses because it deteriorates so quickly. But it gives insight about the forces that hold atoms together so we can learn more about how the universe is held together.
Why are people really doing this? Why do we send particles through huge colliders? Why are we smashing things into each other at higher and higher velocities? I think it fulfills the human race's natural curiosity. We want to know where we come from. And every time we answer something, we come up with ten more questions to answer.
This was a man-made element. How do we know we've found all of the naturally occurring elements?
The good thing with elements is that they're defined by atomic numbers, meaning they're defined by the number of protons in the nucleus. This number is never a fraction, so you can't have, say, 3.2 protons in a nucleus. So we know we have them all because we know of an element with one proton and an element with two protons and so on.
Is there a limit to how many elements we can create?
Well, we're hitting a limit with stability when there are over 90 protons in a nucleus, so while we may find more, we're certainly not getting up to 1,000 protons. It would be too unstable.
One last question: I actually have a periodic table shower curtain. Do you recommend getting an updated one?
I recommend updating your shower curtain when 115 is confirmed. When the committee gets together and names it. And that's an entirely different question.
So I guess I should ask: Why does a committee do it?
Because these things get quite political. Back in the day, the Americans would say: We discovered it and named it something. The Russians would say: We did, and named it something else. So a committee has to get together and negotiate. They try to keep it apolitical-maybe they'll name it after someone from Italy or Lithuania or something.
-GLAIZA GANGAN
In high school science classes, students are encouraged to memorize the first 20 elements of the periodic table. There are other methods employed at the university level for all 118.
So here’s some Method that might be Useful for memorizing The Elements In Periodic Table
How to Memorize the Periodic Table
Uh oh. There's a chemistry test coming up and your teacher wants you to memorize the entire periodic table of the elements. Great. But luckily, with a bit of time and dedication, you can make recalling the table like recalling the alphabet. It'll be as easy as A, B, C. Keep reading for detailed information on how to memorise the periodic table.
Method 1 of 3: The Basics
1. Print out a copy of the periodic table. This will be your Bible for the next couple of weeks. Wherever you go, it will go with you. It's advisable to print out more than one copy. You can highlight and code one however you want and use the next to start over or check if your devices have worked. Print out a copy. Then, especially if you're a visual or kinesthetic learner, copy it down yourself. It's easier to know the ins and outs of something you've done yourself; the chart will seem less foreign if it's made by you.
2. Breakdown the table into smaller sections to learn it. Most charts are already divided by color and type of element, but if that's not working for you, find your own way. You could go by row, column, atomic weight, or simply easiest to hardest. Find patterns that stick out to you.
3. Zap into your free time. Try learning the periodic table when not much else can be done, e.g. traveling by public transport or just waiting in the line for something. If you don't have the chart handy (which you should), go over it in your head, concentrating on the ones that are eluding your memory. Stick with it! Learn a few every day and always review the old ones! If you don't review and quiz yourself, you will forget.
Method 2 of 3: Mind Tricks
1. Create associations. For each element, memorize a short slogan, story or fact that is related to the metal you need to memorize the symbol for. For example, Argentina was named after the metal silver (Argentum -- Ag) because when the Spanish landed there, they thought that the country had lots of silver. Sometimes, you might make something funny to remember the element -- for example," 'EY! YOU! Give me back my GOLD!" could help as well since the symbol for gold is Au.
2. Go for mnemonic devices. That means you'll be using words to associate with each element. They often come in strings or rhymes. LIlly's NAna Kills RuBbish CreatureS FRanticly is an example of a mnemonic device to help remember the alkali metals. Ignore the easy ones. You're probably pretty confident that hydrogen is "H." Concentrate on the ones that are giving you grief. Here's an example: Darmstadtium is "Ds," right? If you want a mnemonic for that one, try "DARN! STATS for my game were all lost on my Nintendo 'DS' because the power went out!".
3. Use pictures. Many people with ridiculously good memories use pictures to associate. Why does everybody know that A is for Apple? Our brains associate words with pictures automatically. Assign each element with a picture -- anything that makes sense to you.
Give the items in your house an element. Label them. Let's say your chair is hydrogen. Label it with a hydrogen bomb, picturing it blowing up. Give your TV a mouth -- it's oxygen and it's breathing. When you go to take your test, close your eyes and walk through your house, recalling all your associations.
4. Memorize in song. If Daniel Radcliffe can do it, so can you.[1] You can either create your own or go on the internet and watch the gems that others have created. If you thought one version is a lot, you'll be pleasantly surprised.
Method 3 of 3: Origins & Patterns, etc.
1. Know the Latin names. All symbols can be regarded as English abbreviations, except for ten that have Latin names and abbreviations and one (Wolfram) whose name can be considered of German origin. Excluding Antimony and Tungsten, these are all important and frequently used elements.Knowing the Latin names as well enables you to decipher most Latin names of inorganic chemicals. In most Romance languages (French, Italian, Spanish, etc.), the present day word is derived from the Latin>
2. Zero in on the differences. Element symbols tend to have two letters. This is the full list of element symbols that have only one letter:
Except for may be V, W and Y, these are all important elements on this table. The symbols D and T (not in this list) are sometimes used for the heavier isotopes of Hydrogen (H). D2O is heavy water.
3.Know which ones come in threes. Elements may have three letters, though. You are probably not required to learn these. These are all highly radioactive, newly discovered (created) elements, that are likely to get new names when the discoveries are confirmed. Professional chemists often don't use these names either, they call it "Element 113" for instance. Just for the heck of it, here is the full list:
4.Spot the unique ones. The last elements that got their names are Flerovium and Livermorium, 114 and 116, whose names were changed from Ununquadium and Ununhexium respectively.
-JERIMAY GARCIA
So here’s some Method that might be Useful for memorizing The Elements In Periodic Table
How to Memorize the Periodic Table
Uh oh. There's a chemistry test coming up and your teacher wants you to memorize the entire periodic table of the elements. Great. But luckily, with a bit of time and dedication, you can make recalling the table like recalling the alphabet. It'll be as easy as A, B, C. Keep reading for detailed information on how to memorise the periodic table.
Method 1 of 3: The Basics
1. Print out a copy of the periodic table. This will be your Bible for the next couple of weeks. Wherever you go, it will go with you. It's advisable to print out more than one copy. You can highlight and code one however you want and use the next to start over or check if your devices have worked. Print out a copy. Then, especially if you're a visual or kinesthetic learner, copy it down yourself. It's easier to know the ins and outs of something you've done yourself; the chart will seem less foreign if it's made by you.
2. Breakdown the table into smaller sections to learn it. Most charts are already divided by color and type of element, but if that's not working for you, find your own way. You could go by row, column, atomic weight, or simply easiest to hardest. Find patterns that stick out to you.
3. Zap into your free time. Try learning the periodic table when not much else can be done, e.g. traveling by public transport or just waiting in the line for something. If you don't have the chart handy (which you should), go over it in your head, concentrating on the ones that are eluding your memory. Stick with it! Learn a few every day and always review the old ones! If you don't review and quiz yourself, you will forget.
Method 2 of 3: Mind Tricks
1. Create associations. For each element, memorize a short slogan, story or fact that is related to the metal you need to memorize the symbol for. For example, Argentina was named after the metal silver (Argentum -- Ag) because when the Spanish landed there, they thought that the country had lots of silver. Sometimes, you might make something funny to remember the element -- for example," 'EY! YOU! Give me back my GOLD!" could help as well since the symbol for gold is Au.
2. Go for mnemonic devices. That means you'll be using words to associate with each element. They often come in strings or rhymes. LIlly's NAna Kills RuBbish CreatureS FRanticly is an example of a mnemonic device to help remember the alkali metals. Ignore the easy ones. You're probably pretty confident that hydrogen is "H." Concentrate on the ones that are giving you grief. Here's an example: Darmstadtium is "Ds," right? If you want a mnemonic for that one, try "DARN! STATS for my game were all lost on my Nintendo 'DS' because the power went out!".
3. Use pictures. Many people with ridiculously good memories use pictures to associate. Why does everybody know that A is for Apple? Our brains associate words with pictures automatically. Assign each element with a picture -- anything that makes sense to you.
Give the items in your house an element. Label them. Let's say your chair is hydrogen. Label it with a hydrogen bomb, picturing it blowing up. Give your TV a mouth -- it's oxygen and it's breathing. When you go to take your test, close your eyes and walk through your house, recalling all your associations.
4. Memorize in song. If Daniel Radcliffe can do it, so can you.[1] You can either create your own or go on the internet and watch the gems that others have created. If you thought one version is a lot, you'll be pleasantly surprised.
Method 3 of 3: Origins & Patterns, etc.
1. Know the Latin names. All symbols can be regarded as English abbreviations, except for ten that have Latin names and abbreviations and one (Wolfram) whose name can be considered of German origin. Excluding Antimony and Tungsten, these are all important and frequently used elements.Knowing the Latin names as well enables you to decipher most Latin names of inorganic chemicals. In most Romance languages (French, Italian, Spanish, etc.), the present day word is derived from the Latin>
2. Zero in on the differences. Element symbols tend to have two letters. This is the full list of element symbols that have only one letter:
Except for may be V, W and Y, these are all important elements on this table. The symbols D and T (not in this list) are sometimes used for the heavier isotopes of Hydrogen (H). D2O is heavy water.
3.Know which ones come in threes. Elements may have three letters, though. You are probably not required to learn these. These are all highly radioactive, newly discovered (created) elements, that are likely to get new names when the discoveries are confirmed. Professional chemists often don't use these names either, they call it "Element 113" for instance. Just for the heck of it, here is the full list:
4.Spot the unique ones. The last elements that got their names are Flerovium and Livermorium, 114 and 116, whose names were changed from Ununquadium and Ununhexium respectively.
-JERIMAY GARCIA
Caffeine, The Greatest Addiction Ever
Every day the world consumes 300 tonnes of caffeine – enough for one cup of coffee for every man, woman and child. The world’s largest buyer of coffee, the US, has to import nearly all of this as the coffee trees from which caffeine is harvested will only grow at commercial levels between the tropic of cancer and the tropic of Capricorn in an area called the coffee belt. Only a single state, Hawaii, is within the belt.
To get this caffeine, first bees must pollinate the flowers of a coffee tree and these flowers develop into bright red berries. Unlike more cooperative domesticated plants, the coffee tree does not ripen all its berries at the same time so they need to be handpicked and sorted. Once picked, the coffee bean is removed from inside the berry. This young seedling of the tree is then dried, heated, ground and submersed in boiling water to get out the precious, precious caffeine. It takes about 40 coffee beans to make one shot of espresso.
But why is caffeine in the coffee beans in the first place? It’s not like the coffee trees want to have humans cutting bits of them off and committing a holocaust of their offspring.
Well, the trees, of course, don’t want or feel anything and originally evolved caffeine for their own benefit. Caffeine is an insecticide that effectively paralyzes or kills bugs chomping on the tree. Whether or not the insects go out experiencing the greatest caffeine high ever is not known.
While caffeine is technically lethal, it’s adapted for for 1g bugs, not monkeys 100,000 times more massive. So you’d really have to try to win this Darwin Award.
But, if you must: to calculate the dose of caffeine you’ll need to ingest to have a 50% of death, take your mass in kilograms and multiply it by 150mg.Or in terms of coffee, for every kilogram of mass you have you need to drink one latte to get a visit from the grim reaper.
That’s a lot of coffee so it’s not surprising that there are no recorded deaths in healthy adults from this method and it’s doubtful that it’s even possible. Because, while you’re busy getting the coffee in, your body is busy getting it out by one way or another.
The rare recorded deaths from caffeine are from diet pills, pep pills and crazy people who eat the drug in its pure form.
Poison though caffeine is, you do still develop addiction to the stuff. And it’s is a real physiological addiction not a wimpy psychological addition like people claim for videos games and the internet.
But caffeine isn’t heroine – rapid withdrawal won’t kill you – it might make you cranky and give you a wicked headache – but since caffeine releases dopamine to make you happy and it gets rid of headaches there’s really no reason to ever stop using it.
And who would want to give up the stuff anyway? I mean, aside from converts to Mormonism and Rastafarianism. Caffeine is the world’s most used psychoactive drug – and with good reason it’s pure awesome.
It increases concentration, decreases fatigue and gives you better memory.
This isn’t just a placebo – these are real effects replicable in a laboratory and, contrary to popular belief, drinking coffee isn’t a Faustian bargain where the devil gives you the ability to work faster but in exchange makes your life shorter. For normal, healthy humans there are no medical concerns. Coffee and the caffeine within it may even has medical benefits such as protection from cardiovascular disease, diabetes and Parkinson’s.
You know what else you can thank caffeine for? A little thing called the enlightenment. In the 1600s people drank more beer and gin than water. But with the introduction of coffee and tea, people switched from a depressant to a stimulant. It’s not surprising then that this time was an intellectual boon compared to earlier centuries.
Ben Franklin and Edward Lloyd loved their coffee for the same reason that modern workers and students do. It’s invaluable for staying awake and concentrating when you need to finish a TPS report or to get through that boring physics class. Coffee is the fuel of the modern world, so go grab a cup guilt-free and get working smarter and faster.
-JULIAN MONTES
Every day the world consumes 300 tonnes of caffeine – enough for one cup of coffee for every man, woman and child. The world’s largest buyer of coffee, the US, has to import nearly all of this as the coffee trees from which caffeine is harvested will only grow at commercial levels between the tropic of cancer and the tropic of Capricorn in an area called the coffee belt. Only a single state, Hawaii, is within the belt.
To get this caffeine, first bees must pollinate the flowers of a coffee tree and these flowers develop into bright red berries. Unlike more cooperative domesticated plants, the coffee tree does not ripen all its berries at the same time so they need to be handpicked and sorted. Once picked, the coffee bean is removed from inside the berry. This young seedling of the tree is then dried, heated, ground and submersed in boiling water to get out the precious, precious caffeine. It takes about 40 coffee beans to make one shot of espresso.
But why is caffeine in the coffee beans in the first place? It’s not like the coffee trees want to have humans cutting bits of them off and committing a holocaust of their offspring.
Well, the trees, of course, don’t want or feel anything and originally evolved caffeine for their own benefit. Caffeine is an insecticide that effectively paralyzes or kills bugs chomping on the tree. Whether or not the insects go out experiencing the greatest caffeine high ever is not known.
While caffeine is technically lethal, it’s adapted for for 1g bugs, not monkeys 100,000 times more massive. So you’d really have to try to win this Darwin Award.
But, if you must: to calculate the dose of caffeine you’ll need to ingest to have a 50% of death, take your mass in kilograms and multiply it by 150mg.Or in terms of coffee, for every kilogram of mass you have you need to drink one latte to get a visit from the grim reaper.
That’s a lot of coffee so it’s not surprising that there are no recorded deaths in healthy adults from this method and it’s doubtful that it’s even possible. Because, while you’re busy getting the coffee in, your body is busy getting it out by one way or another.
The rare recorded deaths from caffeine are from diet pills, pep pills and crazy people who eat the drug in its pure form.
Poison though caffeine is, you do still develop addiction to the stuff. And it’s is a real physiological addiction not a wimpy psychological addition like people claim for videos games and the internet.
But caffeine isn’t heroine – rapid withdrawal won’t kill you – it might make you cranky and give you a wicked headache – but since caffeine releases dopamine to make you happy and it gets rid of headaches there’s really no reason to ever stop using it.
And who would want to give up the stuff anyway? I mean, aside from converts to Mormonism and Rastafarianism. Caffeine is the world’s most used psychoactive drug – and with good reason it’s pure awesome.
It increases concentration, decreases fatigue and gives you better memory.
This isn’t just a placebo – these are real effects replicable in a laboratory and, contrary to popular belief, drinking coffee isn’t a Faustian bargain where the devil gives you the ability to work faster but in exchange makes your life shorter. For normal, healthy humans there are no medical concerns. Coffee and the caffeine within it may even has medical benefits such as protection from cardiovascular disease, diabetes and Parkinson’s.
You know what else you can thank caffeine for? A little thing called the enlightenment. In the 1600s people drank more beer and gin than water. But with the introduction of coffee and tea, people switched from a depressant to a stimulant. It’s not surprising then that this time was an intellectual boon compared to earlier centuries.
Ben Franklin and Edward Lloyd loved their coffee for the same reason that modern workers and students do. It’s invaluable for staying awake and concentrating when you need to finish a TPS report or to get through that boring physics class. Coffee is the fuel of the modern world, so go grab a cup guilt-free and get working smarter and faster.
-JULIAN MONTES
On 10 September 2013 a paper published in Phys. Rev. Lett by D. Rudolph and others outlines additional evidence for the claims that fusion-evaporation reactions between 48Ca and 243Am ions lead to the 288Uup (288115) and 287Uup (287115). A total of thirty correlated α-decay chains were observed following the reactions.
On 12 August 2013 experiments involving zinc ions travelling at 10% of the speed of light colliding with a thin bismuth layer apparently produced a very heavy ion followed by a chain of six consecutive alpha decays identified as products of an isotope of element 113 278Uut - see new evidence for elements 113.
Confirmation of the discoveries of and name proposals for elements 114 (flerovium) and 116 (livermorium)
A news reports from IUPAC (more about flerovium and livermorium) indicates the confirmation of the discoveries of elements 114 and 116:Discovery of the Elements with Atomic Number 114 and 116. Proposals by the discoverers for the names of the two elements have now announced as:
-ERICA BABASA
On 12 August 2013 experiments involving zinc ions travelling at 10% of the speed of light colliding with a thin bismuth layer apparently produced a very heavy ion followed by a chain of six consecutive alpha decays identified as products of an isotope of element 113 278Uut - see new evidence for elements 113.
Confirmation of the discoveries of and name proposals for elements 114 (flerovium) and 116 (livermorium)
A news reports from IUPAC (more about flerovium and livermorium) indicates the confirmation of the discoveries of elements 114 and 116:Discovery of the Elements with Atomic Number 114 and 116. Proposals by the discoverers for the names of the two elements have now announced as:
- element 114: Flerovium (Fl) after the physicist Georgiy Flerov. Georgiy N. Flerov (1913-1990) was a renowned physicist who discovered the spontaneous fission of uranium and was a pioneer in heavy-ion physics.
- element 116: Livermorium (Lv), after the Livermore laboratories. A group of researchers from the Laboratory, along with scientists at the Flerov Laboratory of Nuclear Reactions, participated in the work carried out in Dubna on the synthesis of superheavy elements, including element 116.
-ERICA BABASA
Latest news
On 10 September 2013 a paper published in Phys. Rev. Lett by D. Rudolph and others outlines additional evidence for the claims that fusion-evaporation reactions between 48Ca and 243Am ions lead to the 288Uup (288115) and 287Uup (287115). A total of thirty correlated α-decay chains were observed following the reactions.
On 12 August 2013 experiments involving zinc ions travelling at 10% of the speed of light colliding with a thin bismuth layer apparently produced a very heavy ion followed by a chain of six consecutive alpha decays identified as products of an isotope of element 113 278Uut - see new evidence for elements 113.
Confirmation of the discoveries of and name proposals for elements 114 (flerovium) and 116 (livermorium)
A news reports from IUPAC (more about flerovium and livermorium) indicates the confirmation of the discoveries of elements 114 and 116: Discovery of the Elements with Atomic Number 114 and 116. Proposals by the discoverers for the names of the two elements have now announced as:
element 114: Flerovium (Fl) after the physicist Georgiy Flerov. Georgiy N. Flerov (1913-1990) was a renowned physicist who discovered the spontaneous fission of uranium and was a pioneer in heavy-ion physics. element 116: Livermorium (Lv), after the Livermore laboratories. A group of researchers from the Laboratory, along with scientists at the Flerov Laboratory of Nuclear Reactions, participated in the work carried out in Dubna on the synthesis of superheavy elements, including element 116. Buy Periodic Table posters
Periodic table cartograms poster The Orbitron atomic orbitals poster. You can buy these periodic table posters online and others at our WebElements periodic table shop.
Element 117 discovered?
A paper just published (5 April 2010) in Physical Review Letters by Yu. Ts. Oganessian and others claims the synthesis of a new element with atomic number 117. The abstract states "The discovery of a new chemical element with atomic number Z=117 is reported. The isotopes 293117 and 294117 were produced in fusion reactions between 48Ca and 249Bk. Decay chains involving eleven new nuclei were identified by means of the Dubna Gas Filled Recoil Separator. The measured decay properties show a strong rise of stability for heavier isotopes with Z>=111, validating the concept of the long sought island of enhanced stability for super-heavy nuclei."
-JANE GUEVARRA
-GLAIZA GANGAN
-DANICA TRONGCO
-LANCER GUEVARRA
SOURCE: http://www.webelements.com/
On 10 September 2013 a paper published in Phys. Rev. Lett by D. Rudolph and others outlines additional evidence for the claims that fusion-evaporation reactions between 48Ca and 243Am ions lead to the 288Uup (288115) and 287Uup (287115). A total of thirty correlated α-decay chains were observed following the reactions.
On 12 August 2013 experiments involving zinc ions travelling at 10% of the speed of light colliding with a thin bismuth layer apparently produced a very heavy ion followed by a chain of six consecutive alpha decays identified as products of an isotope of element 113 278Uut - see new evidence for elements 113.
Confirmation of the discoveries of and name proposals for elements 114 (flerovium) and 116 (livermorium)
A news reports from IUPAC (more about flerovium and livermorium) indicates the confirmation of the discoveries of elements 114 and 116: Discovery of the Elements with Atomic Number 114 and 116. Proposals by the discoverers for the names of the two elements have now announced as:
element 114: Flerovium (Fl) after the physicist Georgiy Flerov. Georgiy N. Flerov (1913-1990) was a renowned physicist who discovered the spontaneous fission of uranium and was a pioneer in heavy-ion physics. element 116: Livermorium (Lv), after the Livermore laboratories. A group of researchers from the Laboratory, along with scientists at the Flerov Laboratory of Nuclear Reactions, participated in the work carried out in Dubna on the synthesis of superheavy elements, including element 116. Buy Periodic Table posters
Periodic table cartograms poster The Orbitron atomic orbitals poster. You can buy these periodic table posters online and others at our WebElements periodic table shop.
Element 117 discovered?
A paper just published (5 April 2010) in Physical Review Letters by Yu. Ts. Oganessian and others claims the synthesis of a new element with atomic number 117. The abstract states "The discovery of a new chemical element with atomic number Z=117 is reported. The isotopes 293117 and 294117 were produced in fusion reactions between 48Ca and 249Bk. Decay chains involving eleven new nuclei were identified by means of the Dubna Gas Filled Recoil Separator. The measured decay properties show a strong rise of stability for heavier isotopes with Z>=111, validating the concept of the long sought island of enhanced stability for super-heavy nuclei."
-JANE GUEVARRA
-GLAIZA GANGAN
-DANICA TRONGCO
-LANCER GUEVARRA
SOURCE: http://www.webelements.com/
Periodic Table of Elements The periodic table of elements is a chart that outlines all the basic elements of chemistry that make up our world according to their atomic numbers, the number of electrons each element has, and their predominant chemical properties. Each element is lined up from low to high atomic number, which simply refers to the number of protons it has. Most periodic table of elements charts are laid out in this fashion: A tabular grid of 18 by 7 that houses all of the major elements over another two rows of elements below it. The table can also be broken down into 4 distinct parts or blocks: the s-block on the left, the p-block on the right, the d-block towards the middle and the f-block at the bottom. The table rows are referred to as "periods" and the columns (s, p and d blacks) are called "groups." Some groups also have specific names such as the noble gases, or the halogens. The name "periodic" table suggests that the table itself is open to being updated on a periodic basis, so it's not only used to uncover how each of the elements relate to one another but also to discover the characteristics of new elements or yet to be found or synthesized elements. Therefore, the periodic table proves to be an important guide and resource when it comes to showcasing all the basic elements and studying chemical tendencies, and is commonly used not only in the science of chemistry but other fields of science as well. Although other forms of the periodic table have been known to exist, Dmitri Mendeleev is typically recognized as the pioneer for publishing the first periodic table of elements in 1869. He designed the table to show similarities in the properties of the elements that were known back in the day. He also forecasted the properties of undiscovered elements back then and marked their place on the table, and in fact most of his claims and estimations proved true when the elements were discovered as time passed. Since the 1800's the periodic table has grown and improved with new elements being found and new theories explaining the way chemicals behave. Elements from atomic number 1 to 118, hydrogen to ununoctium, have either been discovered or created. From all of these, all the elements you see up to californium occur naturally. Others have been created in labs. Chemists continue their pursuit to synthesize new elements way beyond ununoctium, but the presence of these synthesized chemicals having their place on the periodic table is still a question of continued disagreement and debate. Synthetic versions of elements that naturally occur in the earth have also been produced in chemical laboratories. Periodic Table of Elements Periodic Table of Elements The above image of an eighteen column period table of elements structure is now the most commonly and broadly used format because it’s been so popular and widely accepted. Also called the “long form” periodic table layout, it differs from Mendeleev’s short form design by removing the groups three to twelve and inserting them instead into the other major groups. The long form layout actually includes the actinides and the lanthanides in its structure instead of placing them below and away from the main table body. This wider layout table also adds two more periods, periods 8 and 9, and also incorporates the superactinides. Periodic Table of Elements One thing to keep in mind is that the periodic table only documents chemical elements. It does not account for subatomic particles, or elements combined together such as mixtures and compounds. Each element's atomic number depends on how many protons it has in its nucleus. Isotopes are two or more variants of the same chemical element. They contain the same number of protons but carry a different number of neutrons in their nucleus. As the number of protons stays equal, the atomic number does not change. For example, carbon has three isotopes that occur naturally. Most have 6 protons but the number of neutrons can vary between 6 and 8. However, isotopes are always shown together under the element they belong to in the periodic table. Elements with no stable isotopes carry the atomic mass of the most stable isotope of that element, where the mass is displayed in parentheses. Let’s take a closer look at the arrangement of elements in the table. As mentioned before, all the elements are arranged according to their rising atomic number, or their increasing number of protons. A new period or row begins when an electron shell gets its first electron. Groups or columns are arranged according to the number of electrons the atom carries. Also, elements that display comparable chemical characteristics usually also fall within the same column, and in the f and d blocks elements that lie in the same row to some extent also display similar characteristics. Therefore if you know the properties of a particular element it is fairly simple to figure out the properties of other elements that surround it in the table. Here are some more facts about the periodic table: According to the most updated version in 2012, the periodic table is said to have 118 elements. Of these 114 are official and have been named and documented by the International Union of Pure and Applied Chemistry (IUPAC). Ninety-eight elements are naturally occurring out of which eighty-four are known as primordial elements. The remaining fourteen occur in decay chains of these primordial elements. Even though elements like livermorium, flerovium and those listed from einsteinium to copernicium do not naturally occur in the earth and have been synthesized, they are still recognized by the IUPAC. Other elements like 113, 115, 117 and 118 are known to be supposedly formulated in labs but the information has not yet been validated. Therefore these elements are only recognized by their element name, depending on their atomic number. As of the year 2012, there have been no reports of any element being synthesized keeping the count as of today at 118.
-DANICA TRONGCO
-JANE GUEVARRA
-GLAIZA GANGAN
-LANCER GUEVARRA
Source: http://www.chemistry2011.org/periodictableofelements
-DANICA TRONGCO
-JANE GUEVARRA
-GLAIZA GANGAN
-LANCER GUEVARRA
Source: http://www.chemistry2011.org/periodictableofelements
